Metabolism of Nitrogen
Metabolism of Nitrogen Nitrogen Cycle Biological Nitrogen Fixation -
We all know that our atmosphere has 78% of the Nitrogen gas which is one of the essential constituent of all living organisms.
Nitrogen cycle is a type of biogeochemical cycle, this term was first coined by Vernadashky in 1934. It consists of two major phases that is biotic phase and the geological phase.
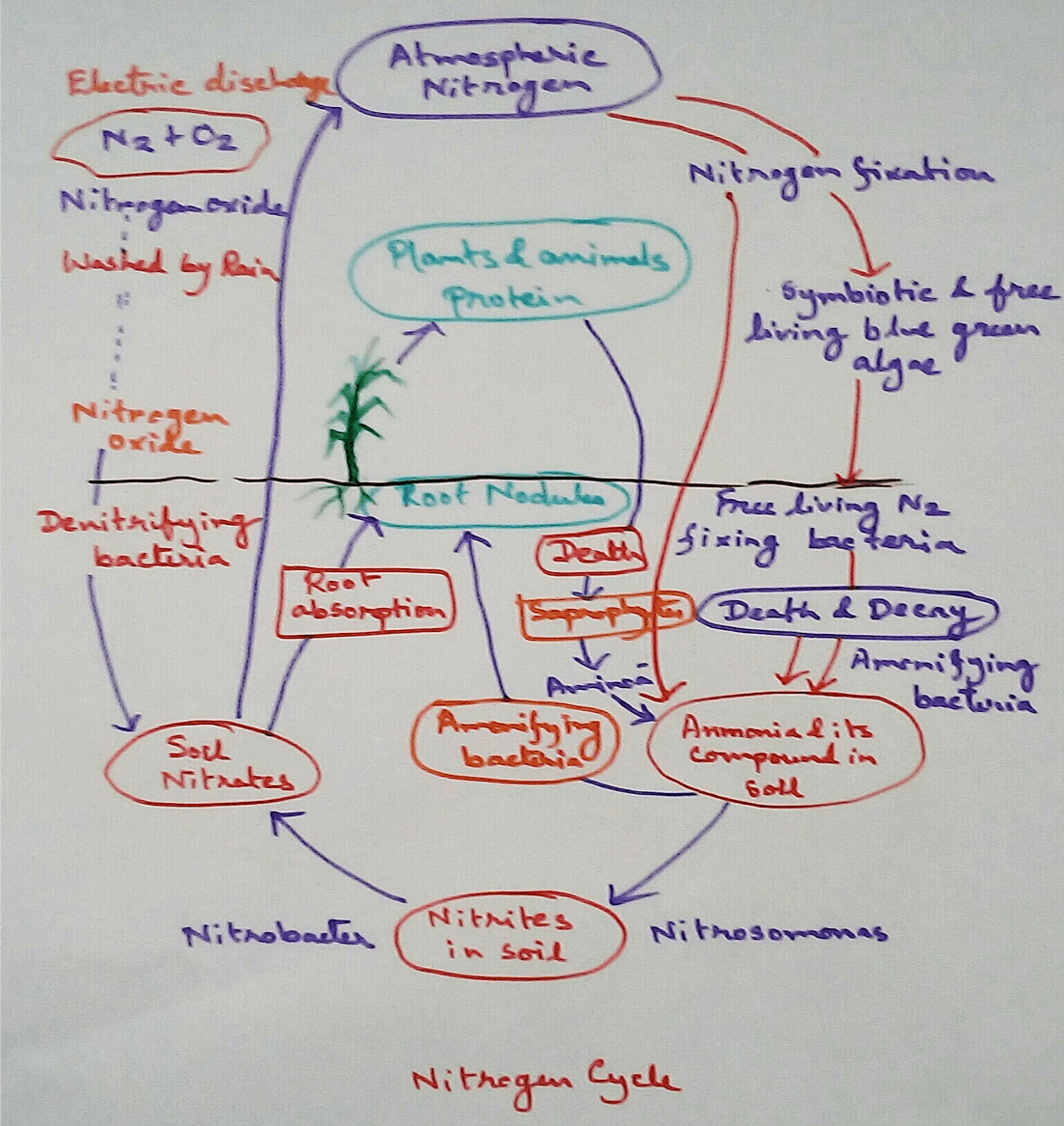
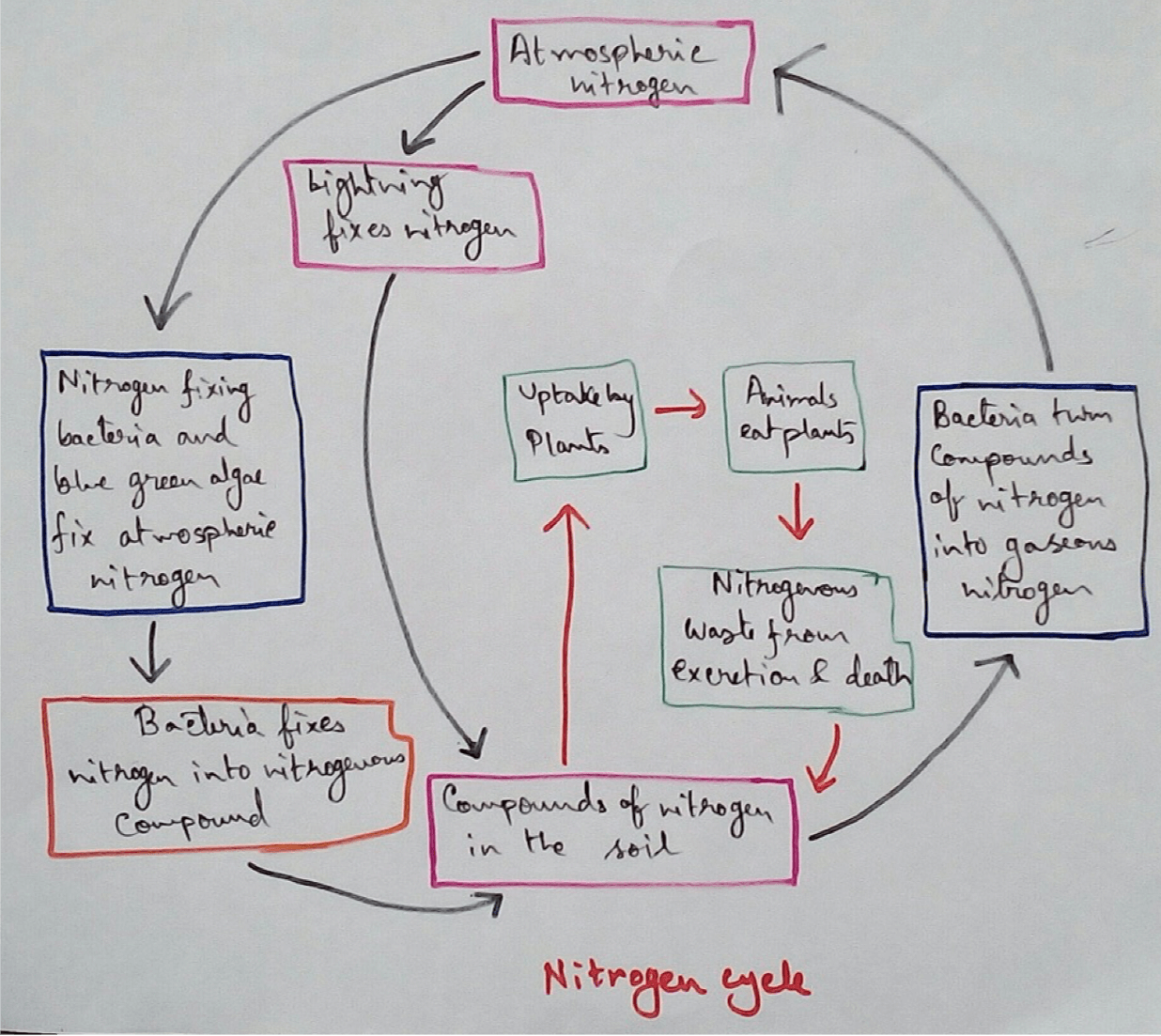
Nitrogen cycles are type of cyclical process for transfer of Nitrogen between the atmospheric nitrogen and soil nitrate take place. Then the atmospheric nitrogen is removed from the atmosphere by the process called nitrogen fixation by free living or symbiotic bacteria.
Significance of Nitrogen - As we all know that protein is a major component of our body which make different enzyme molecules, hormone molecules, biomolecules of DNA, RNA, cell membrane and other organelles. And nitrogen is a major component of protein, so nitrogen is very essential for living beings. Maximum living beings cannot take nitrogen directly from the air though nitrogen occupies maximum percentage in our atmosphere. For this some ammonification, nitrification bacteria are present in the environment that help to nitrogen trap in a nitrate nitrite or Ammonia form. This components from the soil is up taken by the plant by the roots during the absorption of water and are transported to the leaves or other parts of the plants body. Some other type of bacteria are there who are able to denitrifying means they can reduce nitrate component and produce free nitrogen which is been released by them to the environment.
Significance of Nitrogen Cycle - Any bio chemicals cycle in the environment is engaged with the recycling of the component of the atmosphere or environment. As we know when living organisms originate ,its body uptake different elements from the environment and after its death its body decade by different organisms or are mixed with the soil then it is degraded by different microorganisms. This tradition in environment has been created by nature because there is a fixed amount of environmental elements in our surrounding which are uptaken by the organisms during their formation and again returned to the environment with their death. In this returning policy environment is able to maintain its balance for different elements like carbon, water ,Nitrogen, oxygen imoetc.
Biological Nitrogen Fixation - Nitrogen fixation is a process by which we can convert atmospheric free nitrogen to different nitrogenous compound. When this is performed by bacteria and cyanobacteria then it is called biological nitrogen fixation. This biological nitrogen fixation was first discovered in 1891 by the scientist called Winogradsky.. After the conversion of atmospheric nitrogen into different nitrogenous compounds then it can be utilised by plants from the soil through their root system and this nitrogen is used for formation of plant proteins and other compounds then this plant when taken as food by animals this nitrogenous compound enter the animals body.
Type of Nitrogen Fixers - Nitrogen fixers are different type of bacteria those who are able to trapped atmospheric nitrogen in soil nitrate and nitrite form. Nitrogen fixers can be free living or symbiotic.
Free-living Nitrogen Fixation - There are also some free living bacteria called it azotobacter that found in the soil who are able to trap nitrogen from the air as a salt formed. Some blue green algae are also there.
Symbiotic Nitrogen Fixation - Symbiotic nitrogen fixation is observed in rhizobium that lives in the root nodules of the leguminous plants such as beans, Peas and pulses with which it has a symbiotic relationship. Symbiotic relationship is a type of relationship where there is give and take policy applied between two organisms that means someone will give or help second organism where as the second organism will help the first organism. There the root nodules of the pea plants give shelter to the rhizobium bacteria on the other hand rhizobium bacteria are able to trap nitrogen as a nitrate salt or nitrite salt in the soil. Nitrogen cannot up taken by the plants and other living beings directly though it is found maximum percentage in the atmosphere. Thus rhizobium helps the plants to uptake nitrogen by their roots during absorption of water with their minerals and ions formed which is an essential component for the plant.
Structure of Nitrogen Fixing Organisms - Nitrogen fixation requires a specific type of enzyme called Nitrogenase. Beside this type of organisms require ATP a source of hydrogen like nicotinamide adenine diphosphate (NADP)or flavin mononucleotide (FMN) and organic acids for trapping ammonia that produces during nitrogen fixation.
Ammonification or Nitrification - nitrogenous excretion and various nitrogenous substances of dead organisms are acted upon by the decomposers and they produce first amino acids and then ammonia. This entire process is known as ammonification.
Examples are - Bacillus ramosus, B. vulgaris.
Reaction- Proteins + H2O → R – NH2 → Organic Acid + ammonia.
Ammonia is oxidised by nitride bacteria which produce nitrite. Examples are- Nitrosomonas,Nitrosococcus.
Nitrite is there in Father oxidised by nitrate bacteria and fungi to form nitrate. examples of Nitrobacter (bacteria), Penicillium (fungi).
Reaction- N2 + 2H- → 2NH3.
Plants absorb nitrogen from soil and water mostly in the form of nitrates.
Nitrate Assimilation - Nitrogen gas from the environment is trapped as nitrate salt and then it converted to amino acids that are used by the animals and plants.
The nitrate that is observed is reduced by an inducible enzyme called nitrate reductase with the help of N A D (P ) H and FAD /FMN.
Reaction - NO3 - + NAD (P)H + H + - → NO2 - + H2O + NADP +
Nitrite for the reduced by enzyme nitrate reductase with the help of NAD(P)H and ferridoxin which supplies electrons one by one for this reduction and produces ammonia gas.
Reaction - 2NO2 - + 7NAD(P)H + 7H + - → 2NH3 + 4H2O + 7NAD(P) +
The produced Ammonia when combined with different organic acids to form amino acids and then this amino acids are having different combination to form different types of proteins and biomolecules.
From Metabolism of Nitrogen to HOME PAGE
Recent Articles
-
Plants Around Us | Big & Small Plants | Shrubs & Herbs | Water Plants
Feb 03, 26 02:01 AM
We see different types of plants around us. Plants are living things. They breathe and grow. They also reproduce. Most of the plants grow on land. Some plants grow in water. -
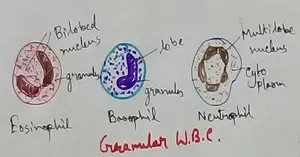
Formed Elements of Blood | Erythrocytes | ESR |Leukocytes |Neutrophils
Jan 15, 26 01:25 AM
Formed elements formed elements are constitute about 45 % of blood afeias haematocrit value packed cell volume mostly of red blood corpuscles and are of 3 types- erythrocytes, leukocytes and blood pla… -
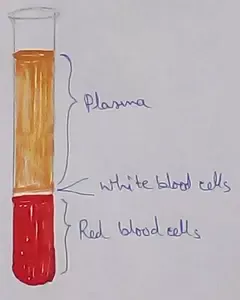
What Is Plasma? | Blood Plasma | Proteins | Nutrients | Cholesterol
Nov 07, 25 10:29 AM
Blood is a mobile fluid which is a connective tissue and is derived from the mesoderm like cell any other connective tissue. Colour of blood is reddish and that flows inside the blood vessels by means… -
Disorders of Respiratory System | Tuberculosis | Pleurisy | Emphysema
Oct 28, 25 11:39 PM
Tuberculosis is very common disease and is caused by a type of bacteria called Mycobacterium tuberculosis. This disease causes different trouble in the respiration and infection of several parts of th… -
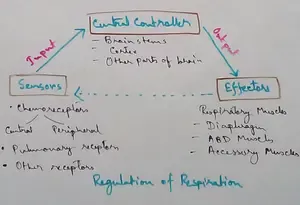
Regulation of Respiration | Respiratory Centres | Inspiratory Area |
Oct 14, 25 12:13 AM
Respiratory Centre is the area that controls the rate of respiration and it is observed to be located in medulla oblongata and pons. Respiratory Centre has the following will dispersed components like…



New! Comments
Have your say about what you just read! Leave me a comment in the box below.