Homogeneous Solution
If there is no particle of solute left undissolved in a solution, the solution is called homogeneous solution. From a homogeneous solution no solute is obtained on filtration. The entire solution passes through the filter paper.
From a homogeneous solution, soluble solute cannot be separated from the solvent by filtration. Solvent and solute can sometimes be separated by heating the solution.
When a pan containing salt solution is heated, the water of the solution evaporates and salt particles are left behind in the pan. In slow evaporation of the water from the solution, the left behind particles of the salt have geometrical shape. These are called the crystals of salt.
When the solution of a substance is placed in a vessel and is allowed to evaporate without heating, the crystals of the solute are obtained after a few days. This way we get the crystals of substances like common-salt, alum, sugar, etc.
When an insoluble substance is dropped in water, it settles down to the bottom of the vessel containing water. The settling down of the substance in the liquid is known as sedimentation. When an insoluble substance settles down to the bottom of the vessel, the clear liquid can be poured slowly into another vessel. This is known as decantation.
From Homogeneous Solution to HOME PAGE
Recent Articles
-
Plants Around Us | Big & Small Plants | Shrubs & Herbs | Water Plants
Feb 03, 26 02:01 AM
We see different types of plants around us. Plants are living things. They breathe and grow. They also reproduce. Most of the plants grow on land. Some plants grow in water. -
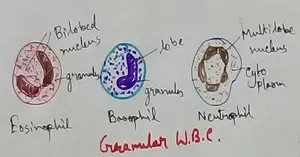
Formed Elements of Blood | Erythrocytes | ESR |Leukocytes |Neutrophils
Jan 15, 26 01:25 AM
Formed elements formed elements are constitute about 45 % of blood afeias haematocrit value packed cell volume mostly of red blood corpuscles and are of 3 types- erythrocytes, leukocytes and blood pla… -
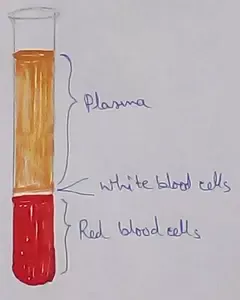
What Is Plasma? | Blood Plasma | Proteins | Nutrients | Cholesterol
Nov 07, 25 10:29 AM
Blood is a mobile fluid which is a connective tissue and is derived from the mesoderm like cell any other connective tissue. Colour of blood is reddish and that flows inside the blood vessels by means… -
Disorders of Respiratory System | Tuberculosis | Pleurisy | Emphysema
Oct 28, 25 11:39 PM
Tuberculosis is very common disease and is caused by a type of bacteria called Mycobacterium tuberculosis. This disease causes different trouble in the respiration and infection of several parts of th… -
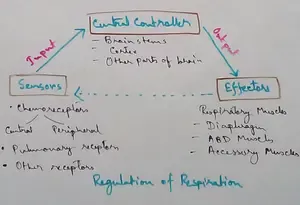
Regulation of Respiration | Respiratory Centres | Inspiratory Area |
Oct 14, 25 12:13 AM
Respiratory Centre is the area that controls the rate of respiration and it is observed to be located in medulla oblongata and pons. Respiratory Centre has the following will dispersed components like…


New! Comments
Have your say about what you just read! Leave me a comment in the box below.