Explain about Cofactors
Definition of Cofactor: Cofactors are organic compound, metallic ions, metallic ion clusters that bind with enzymes and influence the enzyme activity.
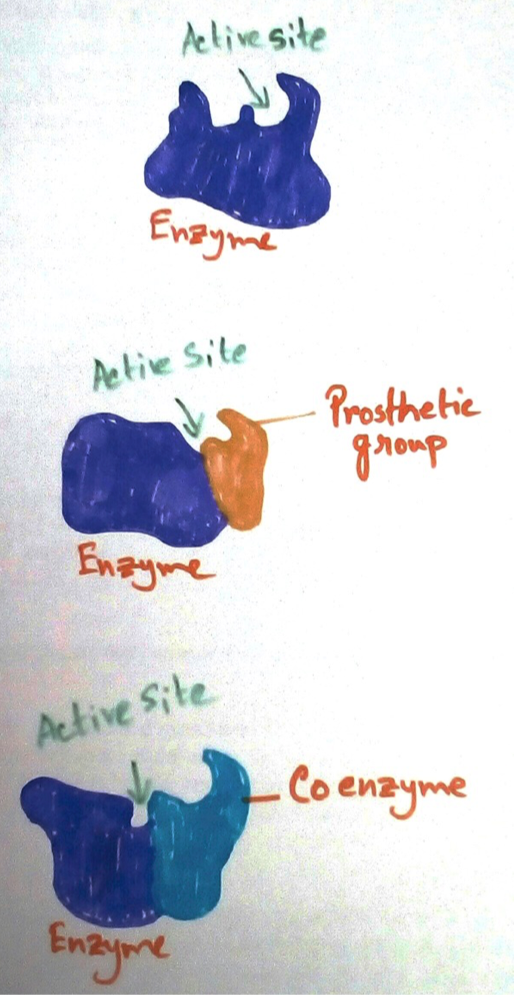
When the cofactor is tightly bound to the enzyme, this is called prosthetic group. If it is loosely bound it is called cofactor. Prosthetic group if binds with the enzyme is a holoenzyme. Protein with holoenzyme is called a holoprotein. Cofactors are covalently bind with the enzymes.
Types of Cofactors: According to the source of origin co factor can be divided into two types broadly. These are –
1. Inorganic Cofactor - Inorganic co factors are mainly metal ions. Metal ions or cluster of metal ions.
Metal Ions – Different metal ions are bind to the enzyme portion and act as co factor. It is already established the importance of trace element in human body for the digestion. Different trace element like – calcium, iron, sulpher, zinc, molybdenum etc act as co factor. Example- Mg ion is used during conversion of glucose to glucose 6 phosphate by glucose 6 phosphatases (enzyme) in glycolysis. Here magnesium ion is used for activation of glucose 6 phosphatases.
Metal Clusters - There are some clusters of metal ions means some metal ions combined together to form metal ions . Example of metal cluster act as co factors is iron -sulpher cluster. It is remain attached with protein amino acid called cysteine. This iron - sulpher cluster act as structural and functional group of redox sensing, electron transfer, structural modules etc.
2. Organic Cofactor - Organic compound which has the molecular weight of less than 1000 Dalton are called organic compound.Different types of organic co factors are vitamin, non vitamin compound etc.
Vitamins as Cofactors - Different vitamins are also bind with the enzyme and help in activation of enzymes.
Example: FAD acts as co factor. Full form of FAD is flavin adenine dinucleotide. It consists of flavin, which is a vitamin, adenine, nucleotide.
Non vitamin co factor- Non vitamin co factors are TPP (thiamine pyro phosphate) acts as co factors of transkrtolase. It is tightly bind with the enzyme and is called prosthetic group. Whereas TPP is loosely bind with the enzyme pyruvate dehydrogenase and is called co factor.
Importance of Cofactors:
First the functions of co factors were remain mysterious. But later some scientist discover their functions . Their functions are-
1. Help in the Transfer of Different Functional Group - Some co factors that remain loosely bind to the enzyme are associated with the transfer of functional group from one compound to the other.
2. Acts as Second Messenger of Hormones - Different hormones binds with their specific receptors and act via some other biomolecules, these biomolecules are called second messenger. They acts on the specific tissues and do their functions. Examples of second messenger are cAMP which binds with G protein couple receptors and help hormone to function.
3. Evolutionary Biology - ATP and NADP are present in almost every living organisms in different structural pattern which help in the study of evolution.
From Factors Affecting Enzyme Activity to HOME PAGE
Recent Articles
-
Plants Around Us | Big & Small Plants | Shrubs & Herbs | Water Plants
Feb 03, 26 02:01 AM
We see different types of plants around us. Plants are living things. They breathe and grow. They also reproduce. Most of the plants grow on land. Some plants grow in water. -
Formed Elements of Blood | Erythrocytes | ESR |Leukocytes |Neutrophils
Jan 15, 26 01:25 AM
Formed elements formed elements are constitute about 45 % of blood afeias haematocrit value packed cell volume mostly of red blood corpuscles and are of 3 types- erythrocytes, leukocytes and blood pla… -
What Is Plasma? | Blood Plasma | Proteins | Nutrients | Cholesterol
Nov 07, 25 10:29 AM
Blood is a mobile fluid which is a connective tissue and is derived from the mesoderm like cell any other connective tissue. Colour of blood is reddish and that flows inside the blood vessels by means… -
Disorders of Respiratory System | Tuberculosis | Pleurisy | Emphysema
Oct 28, 25 11:39 PM
Tuberculosis is very common disease and is caused by a type of bacteria called Mycobacterium tuberculosis. This disease causes different trouble in the respiration and infection of several parts of th… -
Regulation of Respiration | Respiratory Centres | Inspiratory Area |
Oct 14, 25 12:13 AM
Respiratory Centre is the area that controls the rate of respiration and it is observed to be located in medulla oblongata and pons. Respiratory Centre has the following will dispersed components like…




New! Comments
Have your say about what you just read! Leave me a comment in the box below.